Antimicrobial Nanostructures
The global spreading of antibiotic resistance is one of the greatest threats of the 21st century and can be compared to climate change in terms of its severity and effect on the world’s population. In 2019, almost 5 million deaths were associated with drug-resistant bacterial infections. At the same time, however, fewer and fewer new antibiotics are brought to market with yearly FDA approvals steadily decreasing since the 1980s. Without efficient antibiotics, many of the achievements of modern medicine that we today take for granted, such as major surgery, organ transplantation, and cancer chemotherapy, will no longer be available. Therefore, novel and unconventional approaches for the treatment of multidrug-resistant infections are urgently needed. We thus investigate and develop alternative approaches beyond the application of conventional antibiotics to combat drug-resistant bacteria and stop their spreading. In this regard, we focus in particular on the application of nanostructures and nanomaterials.
DNA-based nanoantibiotics
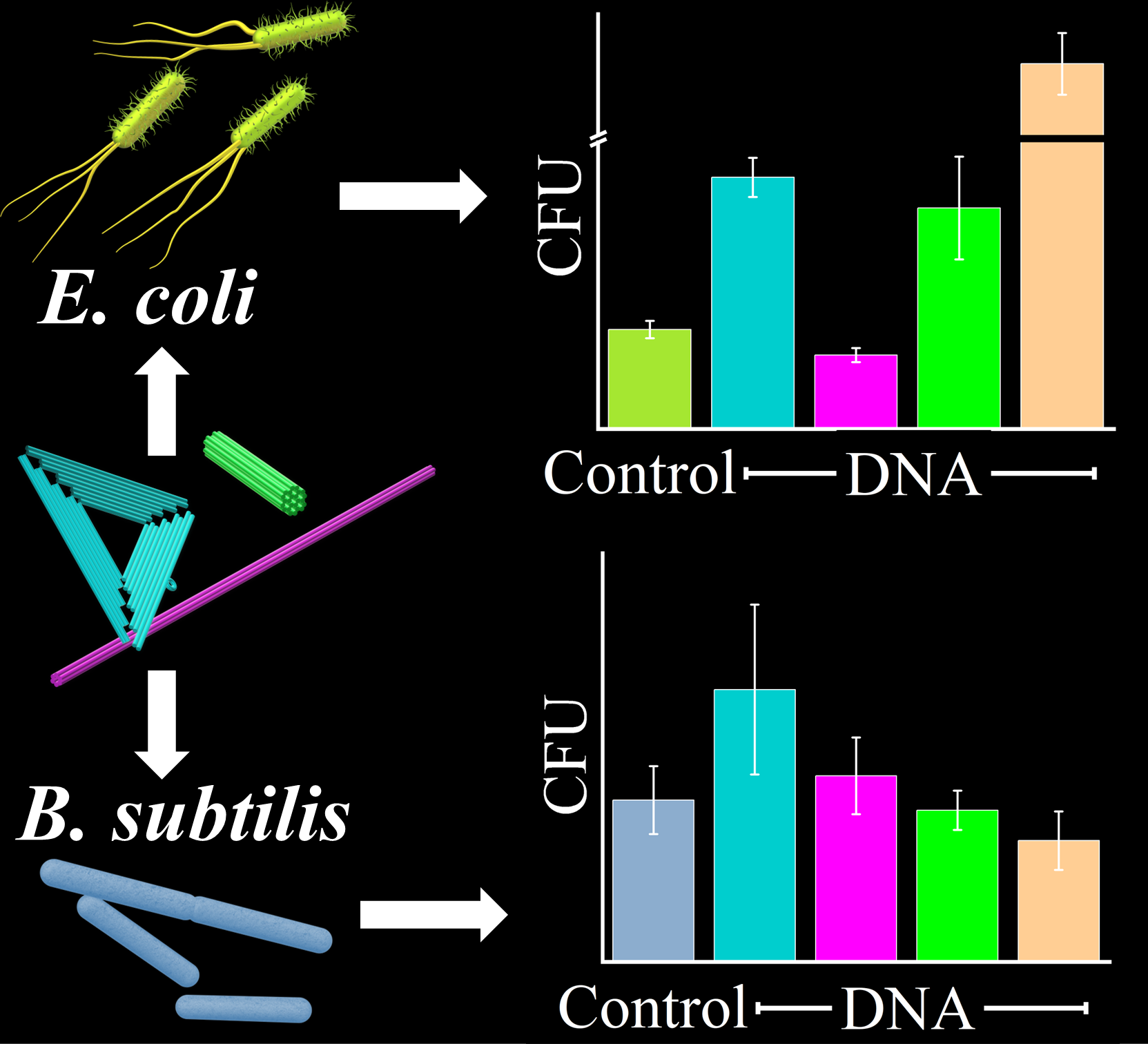
During the last decade, DNA origami nanostructures have become popular and widely employed tools in biomedical research. Compared to conventional organic or inorganic nanoparticles, DNA origami nanostructures have several advantages. (i) Being composed entirely of DNA, they are fully biocompatible and biodegradable, and thus show very low host toxicity. (ii) They can be synthesized with almost arbitrary and highly complex shapes, as well as different mechanical and structural properties, which may be used to tune cellular uptake and biodistribution. (iii) They can be decorated with functional molecules with molecular precision to enable the specific binding of the DNA origami nanostructures to targeted cells. The vast majority of studies in this research area were traditionally aimed at applications in cancer therapy, whereas the field of infectious diseases has largely been neglected. Therefore, our research explores applications of DNA origami nanostructures in antimicrobial drug delivery. We are particularly interested in their controlled loading with antimicrobial molecules and their specific targeting to bacterial pathogens.
Selected publications:
- Effect of DNA Origami Nanostructures on Bacterial Growth, J.A. Garcia-Diosa, G. Grundmeier, and A. Keller, ChemBioChem 25 (2024), e202400091
- Nanoparticle-Based Formulations of Glycopeptide Antibiotics: A Means for Overcoming Vancomycin Resistance in Bacterial Pathogens? B.K. Pothineni and A. Keller, Adv. NanoBiomed Res. 3, 2200134 (2023)
Antimicrobial photodynamic therapy
Antimicrobial photodynamic therapy is a promising alternative to classical antibiotics-based therapeutic approaches. It uses reactive oxygen species (ROS) that are generated by photosensitizers under irradiation with light of a suitable wavelength. These ROS damage surrounding cells through the non-specific oxidation of various cellular subunits, most importantly the cell membrane. While this non-specific mechanism of action makes the emergence of resistance very unlikely, it can also damage host cells. Therefore, we explore new nanotechnology-based strategies to transport photosensitizers directly to the bacterial cells and protect host tissue. Our current research is focused on the application of DNA nanostructures, which on the one hand can efficiently be loaded with established photosensitizers for targeted delivery, but on the other hand may also scavenge and inactivate the generated ROS.
Selected publications:
- Ion-Dependent Stability of DNA Origami Nanostructures in the Presence of Photo-Generated Reactive Oxygen Species, L. Rabbe, J.A. Garcia-Diosa, G. Grundmeier, and A. Keller, Small Struct. 5 (2024) 2400094 (Frontispiece)
- Highly Efficient Quenching of Singlet Oxygen by DNA Origami Nanostructures, J.A. Garcia-Diosa, G. Grundmeier, and A. Keller, Chem. Eur. J. 30 (2024) e202402057 (Cover Picture)